Action Point 8 - Strategy to improve the use and access to reference standards
Last update: 16/07/2025
Background
During the meeting of WG1 Quality in January 2022, the Member States highlighted the challenges they face in accessing reference standards, including for COVID-19, which is critical to test the quality of the products on the market. These concerns were driven by the lack of existing reference standards for some products (e.g. COVID-19 vaccines), but also difficulties in accessing reference standards due to insufficient information about possible sources, challenges in producing reference standards, and the high costs associated with procurement and supply (including transport).
To address this need, the Assembly created in 2022 a dedicated action point (AP8) in the SEARN workplan, led by Working Group 1 (WG1) Quality.
Scope and definitions
General
A reference material is a critical reagent in the developmental cycle of several medicinal products. Among other things, it ensures the products are of high quality and safe. In this document, both chemical and biological reference materials are described. As per the WHO General guidelines for the establishment, maintenance and distribution of chemical reference substances, a chemical reference substance is an ‘authenticated, uniform material that is intended for use in specified chemical and physical tests, in which its properties are compared with those of the product under examination, and which possesses a degree of purity adequate for its intended use’ (1). The same guideline distinguishes:
- Primary chemical reference substance, widely acknowledged to have the appropriate qualities within a specified context, and whose assigned content when used as an assay standard is accepted without requiring comparison with another chemical substance.
- Secondary chemical reference substance, a substance whose characteristics are assigned and/or calibrated by comparison with a primary chemical reference substance.
- Certified reference material (CRS): reference material characterized by a metrologically valid procedure for one or more specified properties, accompanied by a certificate that provides the value of the specified property, its associated uncertainty and a statement of metrological traceability.
- International Chemical Reference Substance, primary chemical reference substances established on the advice of the WHO Expert Committee on Specifications for Pharmaceutical Preparations and supplied primarily for use in physical and chemical tests and assays described in the specifications for quality control of drugs published in The International Pharmacopoeia or proposed in draft monographs.
- Pharmacopeial reference standards, established and distributed by pharmacopeial authorities following the general principles of the ISO Guide: General requirements for the competence of reference material producers (ISO 17034:2016).
Further, the WHO Recommendations for the preparation, characterization and establishment of international and other biological reference standards also identify biological reference standards which are critical for biological medicines and in vitro diagnostics (IVD) (2). Biological reference standards usually comprise materials of complex composition that require biological or immunological assay for appropriate characterization, and are intended to facilitate standardized characterization of biological samples, whatever the type of measurement or method used.
Biological Reference Standards
Biological reference standards have many applications. This material (reagent) can be used to establish a well-defined parameter for assessing safety, efficacy, and quality of IVDs and vaccines. Typically, biological reference materials are generated from convalescent patient samples (blood, plasma, serum, etc.) as they would have shown clinical symptoms of a target disease (in some cases animal model sources are used). Several analytes such as antigens (antibodies and nucleic acids) and/or toxins secreted by the causative agent are assessed in the production of this reagent. Biological reference material can further be refined into international reference standard.
International reference standard (IS) is the primary calibrant and of the highest order. IS are solely established by the WHO Expert Committee on Biological Standardization. Establishment of a WHO IS follows a collaborative study involving various users of the material (including national control laboratories, IVD manufacturers and other accredited laboratories) and as many different, well-established assays as feasible. The material used should resemble as closely as possible the natural analyte of the clinical sample to be measured. An assessment of commutability should be performed as part of the collaborative study where appropriate and feasible. By definition, an IS has a specified value expressed in International Units (IU). This value is arbitrarily assigned based on the results of the collaborative study. WHO IS should not be used for more routine procedures such as validation of assays and as run controls.
Secondary standards (Regional or national reference materials, laboratory or manufacturer’s working calibrator), which is calibrated against the IS. The titre, composition and method of production of secondary standards will vary but should be suitable for obtaining sufficient measurements, when dilution is needed, to achieve an accurate calibration. Regardless of the method of production, each calibration will have a stated measurement uncertainty.
Tertiary standards (external control materials, working reagents or standards, manufacturer’s product calibrator), which is calibrated against the secondary standards. The standard may be formulated from either biological (for example, patient-derived) or non-biological material. However, regardless of the material used, all references in the traceability chain should also demonstrate commutability to the clinical sample of the tested analyte. Tertiary standards are typically formulated as a liquid preparation and may comprise a concentration of the analyte that is detected without dilution in the linear range of the assay it is intended for. They will often be used as an external control material in addition to that normally supplied by the assay manufacturer. Regular monitoring of such material may allow for the early detection of problems with assay performance.
The behaviour of the reference standard should resemble as closely as possible the behaviour of test samples in the assay systems used to test them. As per WHO, ‘The concept of commutability seeks to establish the extent to which the reference standard is suitable to serve as a standard for the variety of samples being assayed’. (TRS 943 - Annex 3: WHO general guidelines for the establishment, maintenance and distribution of chemical reference substances).
General objective
To improve access and use of reference standards in the region.
Guiding framework for the optimal use of reference standards
A Regional hybrid workshop on the optimal use of reference standards was conducted from 14-15 May 2024 in Bangkok, Thailand.
The conclusions of the meeting, endorsed by the Assembly of SEARN, have defined a vision for the regional collaboration between NQCLs:
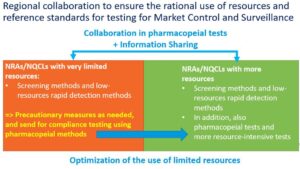
Considering this vision, a set of recommendations were developed, defining a guiding framework for SEARN, WHO and partners which addresses:
- Compliance with good practices and strengthening QMS
- Optimal use of reference standards
- Coordination with other initiatives beyond SEARN.
The details of these recommendations are provided in the highlights from the meeting:
Reducing the use of reference standards
During the 2022-2023 work plan, SEARN also reflected on strategies and good practices to reduce the use of reference standards.
Indeed, procuring reference standards may be difficult in certain areas of the world due to delays in their delivery and the cost of purchase. The number of reference substances prescribed in authoritative analytical procedures shall therefore be reduced, if possible.
Acknowledging this situation, The International Pharmacopoeia applies, for example, the following strategies and practices when elaborating monographs:
- in situ preparation of impurities for identification of related substances/impurities;
- establishment of compounded reference substances that contain several potential impurities for identification;
- quantification of impurities by comparing their detector responses with the response of the parent compound in a diluted sample solution along with the establishment of correction factors to compensate for differences in the responses of the impurity and the parent compound;
- provision of International Infrared Reference Spectra (IIRS) for use in identification tests;
- provision of assay methods not requiring reference substances, like titrations and ultraviolet spectrophotometry using absorptivity values. These methods shall be provided as alternatives in particular to chromatographic assays in monographs for pharmaceutical substances.
These strategies are only be applied when, during the elaboration of the methods, evidence has been obtained demonstrating that the intended measures do not compromise the quality of the analytical results or the ability of the tests to conclusively demonstrate conformance to the applicable standards.
Regional mechanism to list suppliers of reference standards
WG1 Quality was charged with establishing and maintaining a list of trusted suppliers of reference standards. The main mechanism to list suppliers will be reliance (established suppliers, such as pharmacopoeias).
When needed, a regional mechanism for the evaluation of suppliers of reference standards was also developed. WG1 will be responsible for the assessment of new vendors, as well as inspection and testing as required. Based on the assessment, WG1 will provide a recommendation on the inclusion of the new vendor in the regional list.
Reliance on reliable sources of reference standards
As preparation to the list of trusted suppliers of reference standards, SEARN has identified reliable sources of reference standards for chemical and biological reference standards.
Reliable sources of Chemical Reference Standards
The following possible reliable sources of chemical reference standards sources for SEARN NQCLs were identified:
| Organization name | Country | Type |
|---|---|---|
| USP | United States | Primary Reference Standards |
| BP | United Kingdom | Primary Reference Standards |
| Ph.Eur. | France | Primary Reference Standards |
| WHO (Ph.Int.) | France | Primary Reference Standards |
| IPC | India | Primary Reference Standards |
| ASEAN | Thailand | Secondary Reference Standards |
| INCQS | Brazil | Secondary Reference Standards |
| DMSc | Thailand | Secondary Reference Standards |
| NQCLDF | Indonesia | Secondary Reference Standards |
Further links are provided on the WHO website to webpages of pharmacopoeias that provide information on reference substances established to support the performance of analytical provisions described in the respective pharmacopoeias:
Reliable sources of Biological Reference Standards
The following possible reliable sources of biological reference standards sources for SEARN NQCLs were identified:
| Organization name | Country | Type |
|---|---|---|
| Centers for Disease Control and Prevention (CDC) | United States | WHO Biological Reference Standards – custodian laboratories |
| European Directorate for the Quality of Medicines and Healthcare (EDQM) | France | WHO Biological Reference Standards – custodian laboratories |
| National Institute of Allergy and Infectious Diseases (NIAID) | United States | WHO Biological Reference Standards – custodian laboratories |
| National Institute of Biological Standards and Control (NIBSC) | United Kingdom | WHO Biological Reference Standards – custodian laboratories |
| North West Lipid Research Laboratories | United States | WHO Biological Reference Standards – custodian laboratories |
| Paul Ehrlich Institute | Germany | WHO Biological Reference Standards – custodian laboratories |
| Paul Ehrlich Institute | Germany | Secondary reference standards |
| BioQControl | Netherlands | Secondary Reference Standards |
| National Serology Reference Laboratory | Australia | Secondary Reference Standards |
| Royal College of Pathologists of Australasia Quality Assurance Programs (RCPAQAP) | Australia | Secondary reference standards |
Regional list of suppliers of reference standards to support reliance
A regional list of suppliers of reference standards to support reliance was developed, and will be kept up to date if any supplier was assessed:
Regional mechanism to facilitate sharing and testing of samples between the SEARN NQCLs
The possibility of sharing samples between NQCLs is a tool to strengthen the individual capacity for testing in each of the member states, as well as to strengthen the network in terms of a consolidated approach to testing.
Assessment of needs for testing samples outside the NQCL
Due to numerous reasons, such as lack of access to a certain analytical technique and unavailability of reference standards and/or materials, a NQCL may need to have access to another laboratory in which it can rely on for testing.
Also, as part of the market surveillance plan of each of the SEARN members, an exchange programme for planned samples between NQCLs could address the issue of lack of availability of some reference standards and enable a better use and distribution of resources.
The NQCL , herein designated as “interested NQCL”, should clearly define priorities for testing samples in other NQCL, herein designated as “testing NQCL”, such as:
- Samples for routine testing (for instance, for market surveillance purpose), for which there is currently no testing capacity at the NQCL:
- Samples for non-routine testing (for instance, for investigating quality defects or possible substandard and falsified medicines), for which there is currently no testing capacity at the NQCL.
These should be considered as non-exhaustive examples.
SEARN NQCL Capacities database
To support the identification of NQCLs which could provide support for a certain tests, SEARN developed a simple database using an excel spreadsheet, which is shared between all the members of SEARN.
Please refer to AP9 for more information.
Guidance for selecting a testing NQCL and practical aspects to consider
General requirements
Please refer to SEARN AP5 Strategy to facilitate reliance.
Specific requirements
Prior experience with a specific analytical technique, such as gas chromatography coupled with mass spectrometry, dissolution testing or any other analytical technique should be available, if needed.
The testing NQCL should be able to:
- demonstrate prior experience with the product to be tested, or be able to prepare the testing prior to receiving the sample,
- perform the method verification, or if applicable, the method validation;
- inform about the minimum amount of sample required for the testing requested.
The testing NQCL should be able to provide a date for issuance of the final test report / certificate of analysis.
Ad hoc testing
Based on the priorities established, the NQCL should prepare a call for Expression of Interest (EoI) to any potential testing laboratory, preferably another NQCL, clarifying which are the testing requirements, such as:
- which are the tests to be performed,
- which is the testing method to be used – if applicable,
- number of samples to be tested,
- transport and storage requirements for the samples,
- timelines for the delivery of results,
- and any other testing requirements.
The bidding process should also be detailed and clarified in the EoI, as well as specific financial details and timelines for submission of proposals.
Information about specific custom requirements to be expected for the shipping of samples and entry should also be requested to the bidder(s).
Systematic testing
In order to facilitate a request for testing, interested NQCLs can also establish MoU or contracts with another NQCL , the testing NQCL”), to perform specific testing on their behalf.
The requirements of this agreement may include the information detailed in sections “Specific requirements” and “Ad-hoc testing”, but should address specifically the needs of the interested NQCL. Information on who is responsible for certain aspects, such as acquiring reagents, materials, reference standards, costs for shipping the samples and customs fees as well as maximum number of samples to be tested per period of time, should also be addressed, for transparency purposes.
NQCLs are encouraged to address this possibility with their top management, to determine feasibility and acceptability, as well as to establish the maximum capacity that the NQCL is able to provide to other NQCLs in general terms and for a specific period of time.
Financial aspects
Ideally, an exchange programme between NQCLs, with a very limited number of samples to be tested, for instance, under the testing NQCL national market surveillance programme, would be cost free, with the exception of the shipping and customs fees. However, this situation will configure more the exception than the rule.
Hence, the interested NQCL should ensure that funding is available to support testing, if this is required in any MoU or contract. When national funding is not available, support from development partners or WHO could be explored.
Provisions should be in place to ensure that the interested NQCL is able to perform the payment in the currency required by the testing NQCL, as described in the terms of the signed MoU or contract. However, challenges are expected to arise at this level, as national requirements for currencies used for this purpose may not be acceptable both for the interested parties, and this should be clearly discussed.
Alignment between NQCL needs and production of reference standards from established regional and international producers and pharmacopoeias
Engagement with the International Meetings of World Pharmacopoeias
In order to optimize the use of reference standards and facilitate access for SEARN members, SEARN engaged the main pharmacopoeias by participating in the 15th International Meetings of World Pharmacopoeias, New Delhi, India, 6- 7 February 2025.
During the meeting, the Chair of WG1 Quality highlighted that countries without pharmacopoeias often have less resources but require to spend much more on reference standards as manufacturers refer to different pharmacopoeias.
He advocated for:
- reduced use of RS in monographs
- enabling use of RS from different pharmacopoeias
- Develop / communicate access schemes for NQCLs
During IMWP, the participants representing world pharmacopoeias ‘noted the importance of finding a mechanism to hear and consider the perspectives of pharmacopoeias not participating in IMWP as well as those of countries without a pharmacopoeia’.
SEARN agreed to continue engaging with IMWP in the future.
Engagement with producers of reference standards
In order to support its engagement with producers of reference standards and identify and advocate for access programmes for NRAs, SEARN decided to conduct a series of interviews.
In order to support this plan of actions, a discussion guide was developed.
Support Members in their risk-based approaches through identifying minimum standards for screening strategies
SEARN identified and prioritized the main needs for which there is a need for developing risk-based approaches through identifying minimum standards for screening strategies as follows:
- Sterility
- Identification of sildenafil citrate
- Identification of dexamethasone
- Identification of amoxicillin
- Identification of ciprofloxacin
- Identification of clonazepam
In general, with respect to active substances, a general high-level strategy was proposed, which would require support from a laboratory willing to support the development of these methods:
- TLC method for identification
- HPLC to quantify if suspicion
Biological reference standards
A survey was conducted to identify the needs for biological reference standards in SEARN countries.
Highlights:
- About half of SEARN members indicated that their laboratory has the capacity to conduct tests requiring biological reference standards. When it was not the case, several members indicated intentions to develop this capacity.
- SEARN members indicated that:
- they mostly require biological reference standards for vaccines/biological medicines/IVDs for screening and confirmatory tests for market control and surveillance, and marketing authorization, which would require further discussion.
- A few members also indicated needs related to lot release and export.
- Members were also requested to rank the top 5 biological reference standards they required for testing biological medicines, vaccines, and IVDs. While rabies vaccines and anti-rabies immunoglobulin slightly stood out, this part of the survey could not elicit clear shared needs.
- Challenges related to accessing biological reference standards also appeared diverse:
- Only a minority of respondents selected each proposed challenge.
- Costs was the most identified challenge (3 responses), followed equally (2 responses) by the absence of supplier, custom clearance and delays in receiving the reference standards.
Based on these results, SEARN considered that further in-depth discussion on the need for biological reference standards and their use was required. These discussions will also provide an opportunity to better consider biological reference standards in the strategy on access to reference standards, which was mostly developed considering chemical reference standards.
Next steps
- To continue the engagement with the International Meetings of World Pharmacopoeias
- To conduct interviews with the identified reliable producers of reference standards
- To identify or develop risk-based approaches for prioritised tests through identifying minimum standards for screening strategies
- To conduct one or several workshops to better characterise the needs of SEARN members regarding biological reference standards and define the related strategy of the Network
References
- Avumegah, M.S., Mattiuzzo, G., Särnefält, A. et al. Availability and use of Standards in vaccine development. npj Vaccines 8, 95 (2023). https://doi.org/10.1038/s41541-023-00692-0
- General guidelines for the establishment, maintenance and distribution of chemical reference substances - TRS 943 - Annex 3 [Internet]. 2007. Available from: https://www.who.int/docs/default-source/medicines/norms-and-standards/guidelines/quality-control/trs943-annex3-establishmentmaintenance-distribution-chemica-reference-substances.pdf?sfvrsn=71064286_0
- Recommendations for the preparation, characterization and establishment of international and other biological reference standards, Annex 2, TRS No 932 [Internet]. 2004. Available from: https://www.who.int/publications/m/item/annex2-trs932
- WHO manual for the preparation of secondary reference materials for in vitro diagnostic assays designed for infectious disease nucleic acid or antigen detection: calibration to WHO International Standards, Annex 6, TRS No 1004. https://cdn.who.int/media/docs/default-source/biologicals/blood-products/document- migration/secstandmanwho_trs_1004_web_annex_6.pdf?sfvrsn=d6937af8_3&download=true
